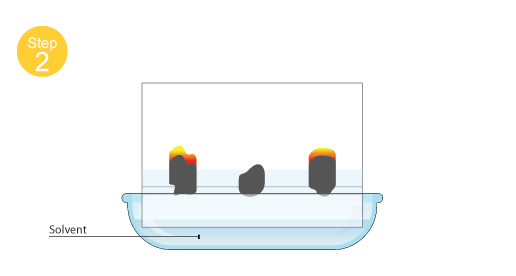
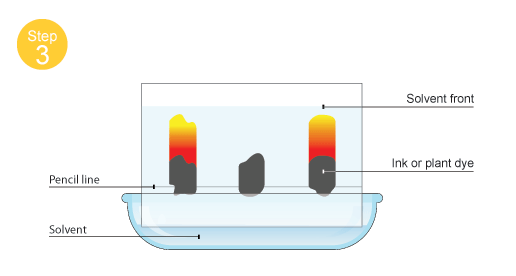
C2. Experimental techniques
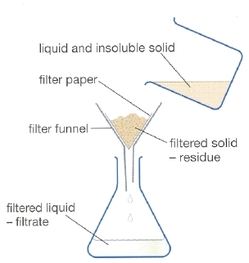
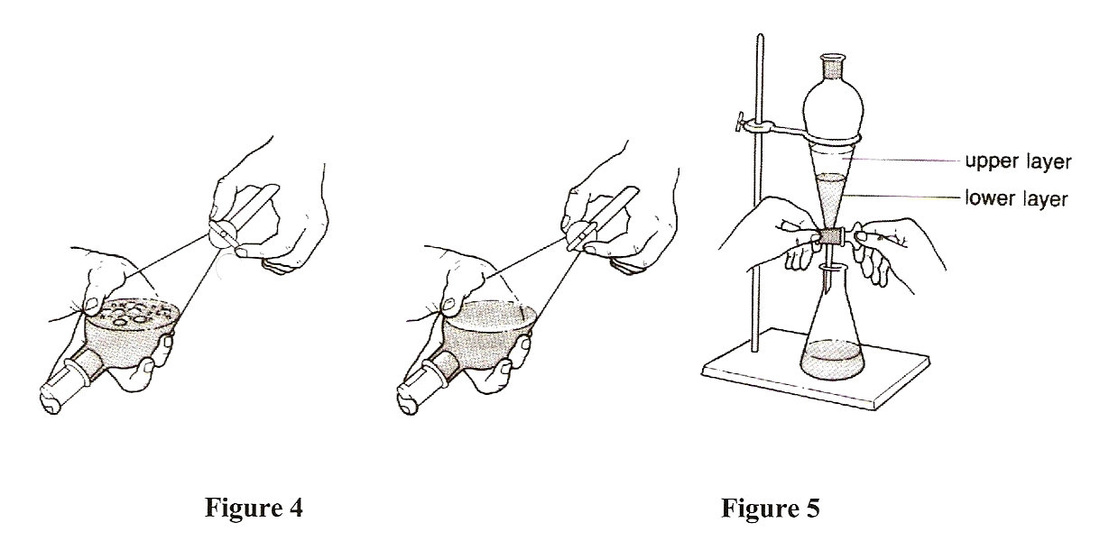
Filtration- Solid/Liquid
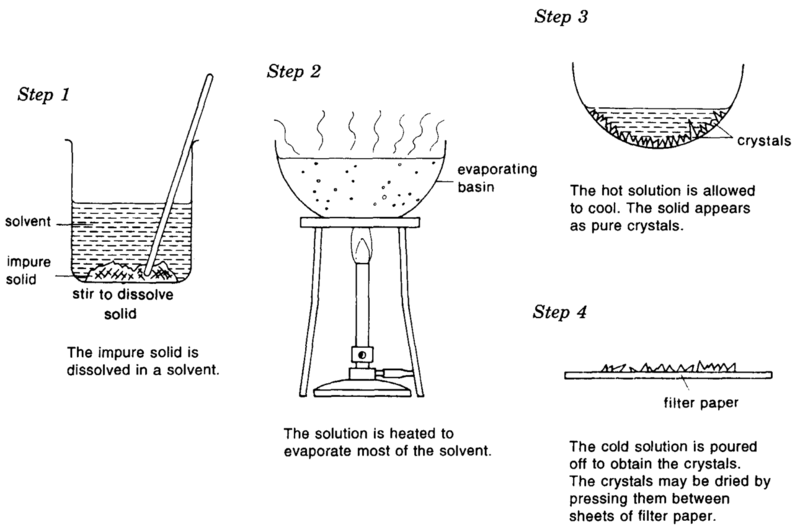
Crystallisation- Pure solid sample from its solution
- Heat the solution until most of the solvent has evaporated off. A hot saturated solution is obtained.
- Allow the saturated solution to cool. The solute now appears as pure crystals while impurities stay in the solution.
- Pat the crystals dry with filter paper to obtain a pure dry crystals.
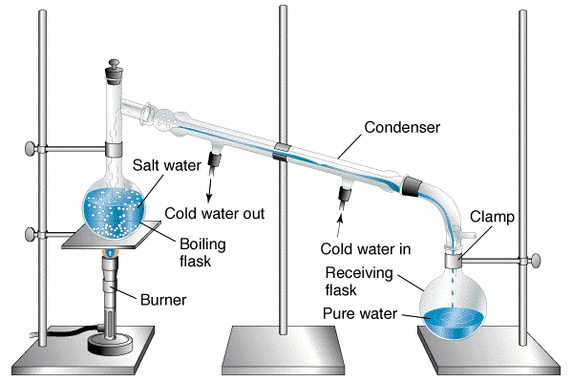
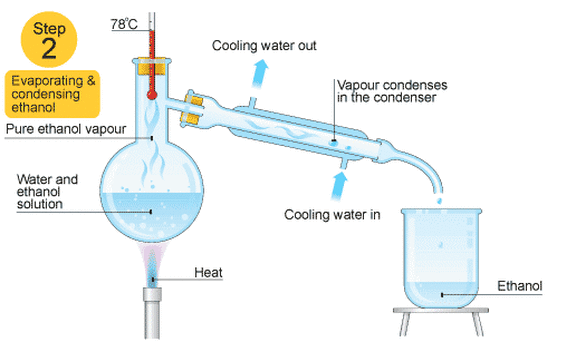
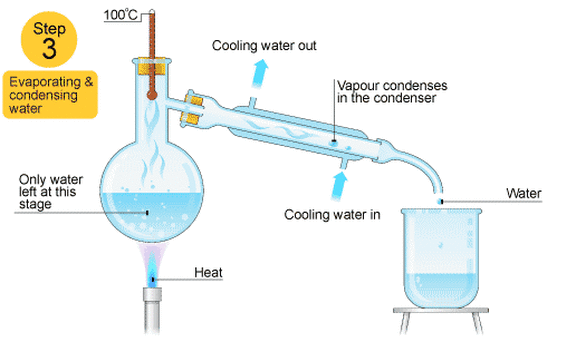
Distillation- Solid/Liquid solution
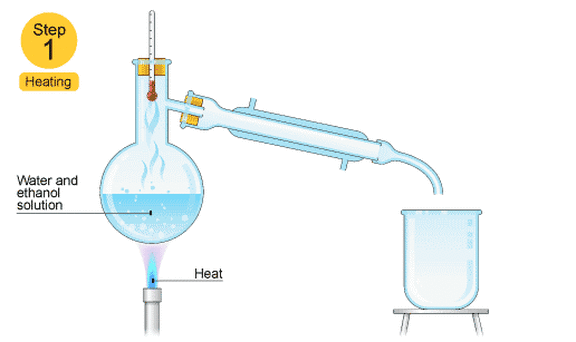
This is good for separating two or more liquids from each other. For example, ethanol (alcohol) can be separated from a mixture of ethanol and water by fractional distillation. This method works because the two liquids have different boiling points.