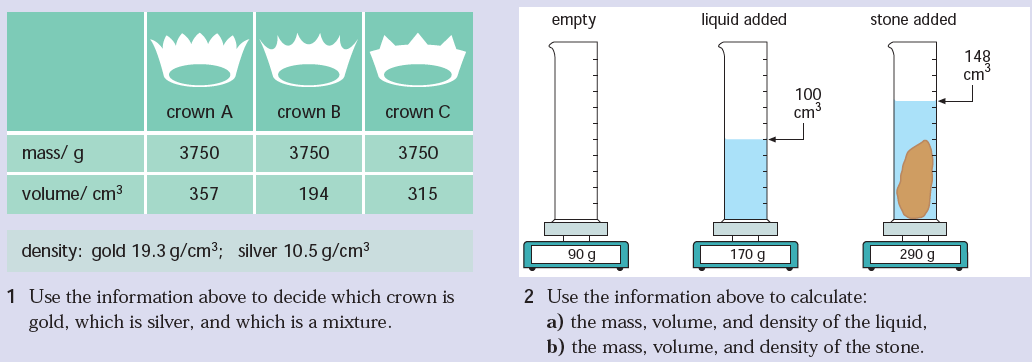
P13. Density
|
|
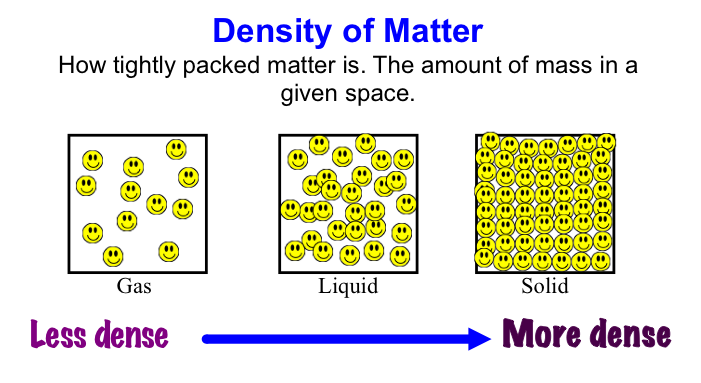
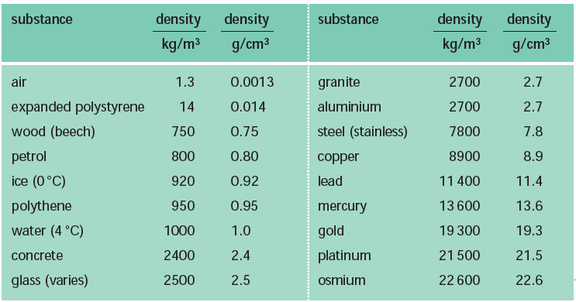
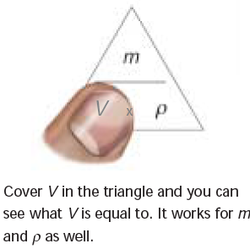
Density is a physical property of a substance. At any given temperature and pressure the density of a material is constant. Density is related to two attributes: mass and volume. This gives rise to the formula:
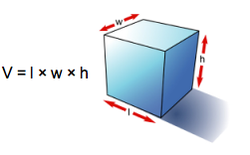
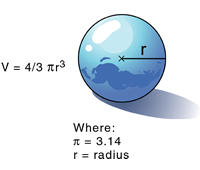
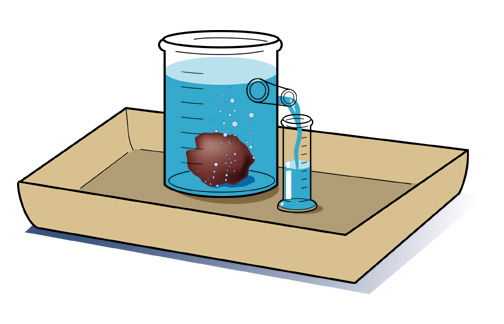
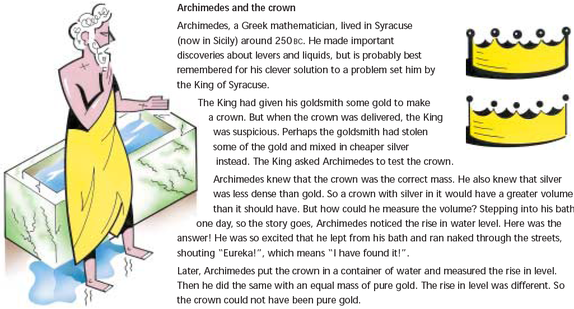
In a laboratory it is possible to determine the mass of an object using a balance. It is also easy to determine the volume of a material; for example, a graduated cylinder can be used to find the volume of a liquid. The volume of a regularly shaped solid can be determined mathematically. It gets a little trickier to determine the volume of an irregular solid; you have to use displacement of water (Archimedes Principle) to find the volume.
Density is typically expressed in grams / cubic centimeter (g/cm3). Equal volumes of different liquids do not necessarily have the same mass. You can demonstrate this by setting up the following situation. All you need are equal volumes of water and corn oil (less dense than water) or corn syrup (more dense than water). The volume of an irregular solid, such as a stone, can be determined by displacement of water. Fill an overflow can so that the water is just above the level of the spout and plug the spout with your finger. Place the overflow can on the counter with its spout above a sink or put the can in a catch basin, uncover the spout and allow the excess water to flow out. Now position a graduated cylinder under the over-flow spout to catch the water that will flow out. Carefully drop the irregularly shaped object into the can and catch the overflowing water in the cylinder. Read the volume of water collected in the graduated cylinder using the markings on the side. The volume of water in the cylinder is equal to the volume of the object. |