Chapter 8 & 10: The periodic table and reactivity
|
There are more than 100 different elements. The periodic table is a chart showing all the elements arranged in a particular way. The vertical columns in the periodic table are called groups. Each group contains elements that have similar properties.
All the different elements are arranged in a chart called the periodic table.
|
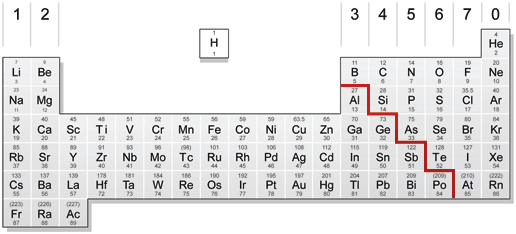
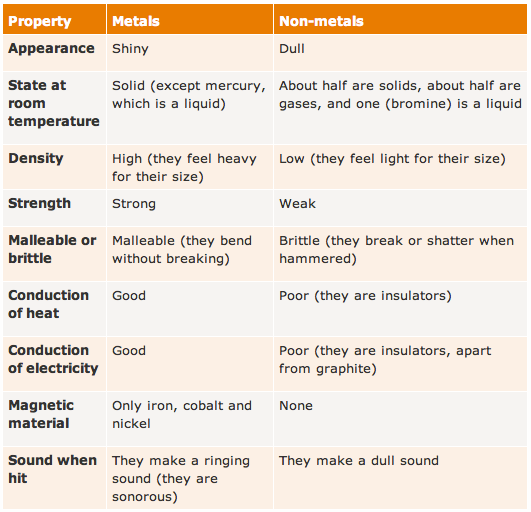
The zig-zag line in this diagram separates the metals, on the left, from non-metals, on the right. Hydrogen is a non-metal but it is often put in the middle.
Notice that most elements are metals, rather than non-metals.
Notice that most elements are metals, rather than non-metals.
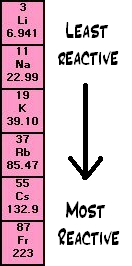
Group 1- The alkali metals
The elements in group 1 are called the alkali metals. They belong to the left-hand column in the periodic table. They are very reactive and must be stored in oil to avoid contact with air or water.
Common properties
The alkali metals have the following properties in common:
- they have low melting and boiling points compared to most other metals
- they are very soft and can be cut easily with a knife
- they have low densities (lithium, sodium and potassium will float on water)
- they react quickly with water, producing hydroxides and hydrogen gas
- their hydroxides and oxides dissolve in water to form alkaline solutions
Reactions of lithium, sodium and potassium with water
The hydroxides formed in all these reactions dissolve in the water to form alkaline solutions. These turn universal indicator purple - showing that the pH is more than 7.
|
|
Group 2- The alkaline earth metals
These elements are all found in the Earth’s crust, but not in the elemental form as they are so reactive. Instead, they are widely distributed in rock structures. The main minerals in which magnesium is found are carnellite, magnesite and dolomite. Calcium is found in chalk, limestone, gypsum and anhydrite. Magnesium is the eighth most abundant element in the Earth’s crust, and calcium is the fifth.
Common properties
The alkaline earth metals have the following properties in common:
- they have higher melting and boiling points compared to the alkali metals
- they are quite soft but are harder than the alkali metals
- they have greater densities than the alkali metals
- they react less vigorously with water
- their hydroxides and oxides dissolve in water to form alkaline solutions
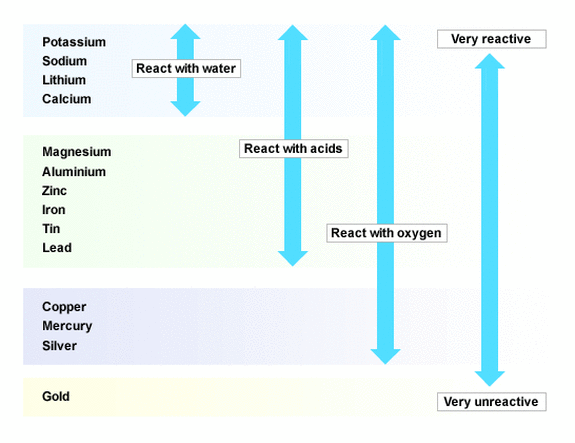
Trends in reactivity
As you go down the group, the atomic number of the alkali metals increases, and their properties change:
- their melting points decrease (except for Mg is anomalously low)
- their densities decreases
- they become softer
- they become more reactive
Reactions of alkaline earth metals (Mg & Ca) with water
The hydroxides formed in all these reactions dissolve in the water to form alkaline solutions. These turn universal indicator purple - showing that the pH is more than 7. The reactions are seen to be less vigorous than with the Group 1 alkali metals.
|
|
|
Reactions of alkaline earth metals (Mg & Ca) with hydrochloric acid (HCl)
Magnesium and calcium react readily with dilute hydrochloric acid to form hydrogen gas and a water soluble metal salt. Like group 1, one observes an increase in reactivity down the group (i.e. Ca is more reactive than Mg)
|
|
|
Transition metals
The transition metals are found between group 3 & 4 on the periodic table.
They comprise:
21 (Scandium) through 29 (Copper)
39 (Yttrium) through 47 (Silver)
57 (Lanthanum) through 79 (Gold)
89 (Actinium) and all higher numbers.
The transition metals have the following key features:
- they are good conductors of heat and electricity
- they are often found freely in nature
- they are malleable (can be bent or hammered into shape)
- Useful for structural materials
Group 7- The halogens
The elements in group 7 are called the halogens. They belong to the column second from right in the periodic table. The halogens are all toxic, but this is a useful property. Chlorine is used to sterilise drinking water and water in swimming pools. Iodine is used in antiseptics to treat wounds.
Common properties
The halogens have the following properties in common:
- they are non-metals
- they have low melting and boiling points
- they are brittle when solid
- they are poor conductors of heat and electricity
- they have coloured vapours
- their molecules each contain two atoms (they are diatomic)
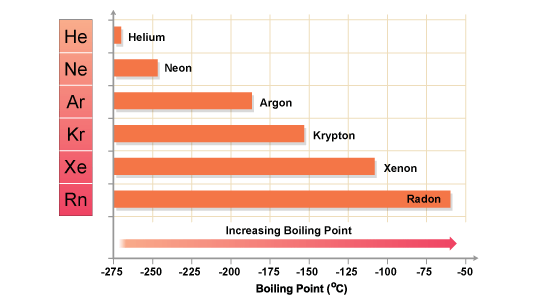
The halogens have low melting points and boiling points. This is a typical property of non-metals. You can see from the graph that fluorine, at the top of Group 7, has the lowest melting point and boiling point in the Group. The melting points and boiling points then increase as you go down the Group.
Group 0- The noble gases
The elements in group 0 are called the noble gases. They belong to the right-hand column in the periodic table. The noble gases are all chemically unreactive which means they are inert.
Common properties
The noble gases have the following properties in common:
- they are non-metals
- they are very unreactive gases
- they are colourless
- they exist as single atoms (they are monatomic)
Physical properties
|
|
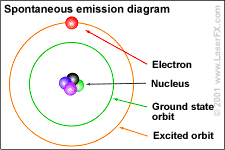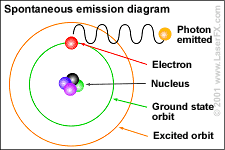
When an electrical charge travels through the gas in the tube (energy is pumped into the gas), it excites or stimulates the atoms. Some of the electrons absorb this energy by jumping up to the next highest orbit.
This configuration is unstable. The electron wants to return to its regular orbit, the ground state. As the excited (stimulated) atoms in the gas relax back to the ground state, some of the energy that excited the electron(s) is emitted (released) in the form of random photons of light (see diagram to the left). |