C6. Energy changes in chemical reactions
Here’s the basics: when chemical reactions occur, energy is transferred (because energy is required to break and make bonds). Two things can happen in this case: either the reaction ‘gives out’ heat (energy) to its surroundings, or it ‘takes’ heat (energy) from its surroundings. We have two special terms for these events:
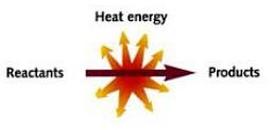
Exothermic - This is when the reaction ‘gives out’ energy, in the form of heat. Think of it as heat exiting the reaction (or alternatively, just learn the two different definitions – there are only two after all…
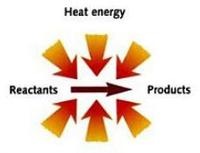
Endothermic - You guessed it – this is when the reaction ‘takes’ (usually heat) energy from the surroundings, for its own devious purposes. You could think of it as energy entering the reaction.
Exothermic - This is when the reaction ‘gives out’ energy, in the form of heat. Think of it as heat exiting the reaction (or alternatively, just learn the two different definitions – there are only two after all…
Endothermic - You guessed it – this is when the reaction ‘takes’ (usually heat) energy from the surroundings, for its own devious purposes. You could think of it as energy entering the reaction.
Examples of Exothermic Reactions
Chances are, you won’t directly be asked a question that says: ‘state a type of exothermic reaction’ - however, you may be asked to talk about a reaction that is exothermic! With this in mind, here are some examples of exothermic reactions:
Chances are, you won’t directly be asked a question that says: ‘state a type of exothermic reaction’ - however, you may be asked to talk about a reaction that is exothermic! With this in mind, here are some examples of exothermic reactions:
- Combustion reactions – burning things!
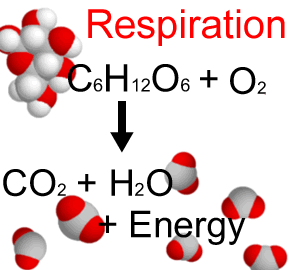
- Oxidation reactions – a good example of this is respiration: this is why respiring ‘gives out’ heat (a good fact to know for biology!)
- Neutralisation reactions – reactions that involve acids and bases – making some salts causes this!
An Example of An Endothermic Reaction
In the same way as above, you may have to talk about an endothermic reaction.
The main candidate for an endothermic reaction is a thermal decomposition reaction. This type of reaction is where a substance breaks up into two or more separate parts; it’s endothermic because it requires a constant input of heat for the decomposition to continue. The only other major type of reaction that is endothermic is occasionally when you dissolve some solid compounds into water. The temperature of the water decreases as some endothermic changes happen in the dissolving process.
In the same way as above, you may have to talk about an endothermic reaction.
The main candidate for an endothermic reaction is a thermal decomposition reaction. This type of reaction is where a substance breaks up into two or more separate parts; it’s endothermic because it requires a constant input of heat for the decomposition to continue. The only other major type of reaction that is endothermic is occasionally when you dissolve some solid compounds into water. The temperature of the water decreases as some endothermic changes happen in the dissolving process.