P5. Matter and Thermal Properties
5.1 Thermal expansion of solids, liquids and gases
|
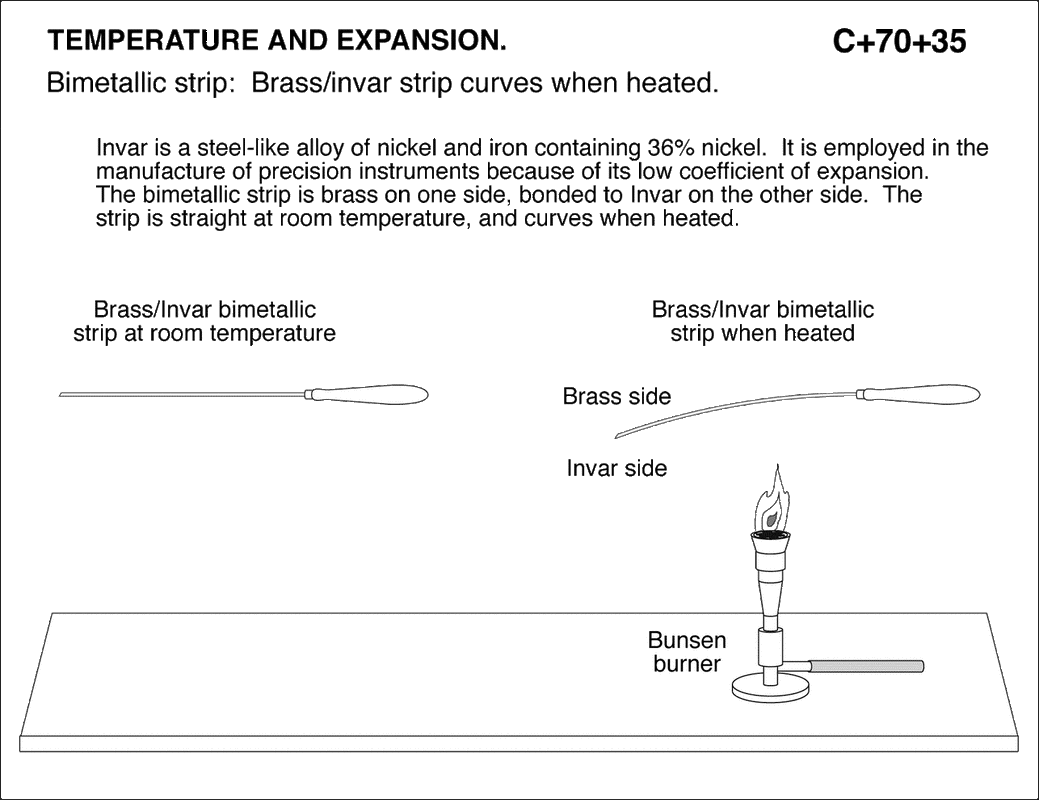
Thermal Expansion and Contraction of a Solid
|
|
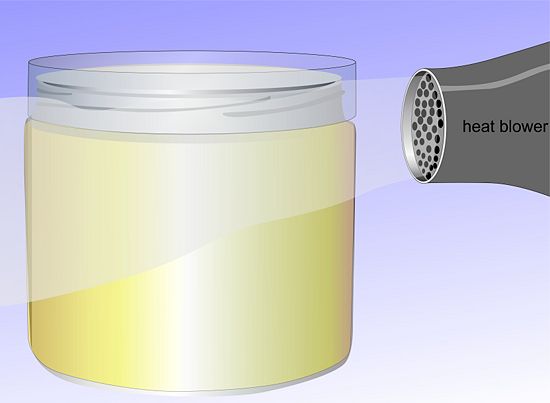
Thermal Expansion of a Liquid in a round-bottom flask
|
|
Helium gas contracting and expanding in a mylar balloon
|
|
Thermal Expansion - Real-life applications
Opening Jars
An everyday example of thermal expansion can be seen in the kitchen. Almost everyone has had the experience of trying unsuccessfully to budge a tight metal lid on a glass container, and after running hot water over the lid, finding that it gives way and opens at last. The reason for this is that the high-temperature water causes the metal lid to expand.
An everyday example of thermal expansion can be seen in the kitchen. Almost everyone has had the experience of trying unsuccessfully to budge a tight metal lid on a glass container, and after running hot water over the lid, finding that it gives way and opens at last. The reason for this is that the high-temperature water causes the metal lid to expand.
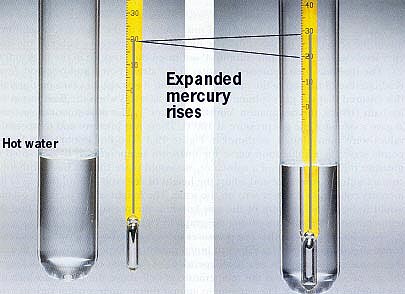
Thermometers
In a typical mercury thermometer, mercury is placed in a long, narrow sealed tube called a capillary. Because it expands at a much faster rate than the glass capillary, mercury rises and falls with the temperature. A thermometer is calibrated by measuring the difference in height between mercury at the freezing point of water, and mercury at the boiling point of water. The interval between these two points is then divided into equal increments in accordance with one of the well-known temperature scales.
In a typical mercury thermometer, mercury is placed in a long, narrow sealed tube called a capillary. Because it expands at a much faster rate than the glass capillary, mercury rises and falls with the temperature. A thermometer is calibrated by measuring the difference in height between mercury at the freezing point of water, and mercury at the boiling point of water. The interval between these two points is then divided into equal increments in accordance with one of the well-known temperature scales.
Expansion Joints
Most large bridges include expansion joints, which look rather like two metal combs facing one another, their teeth interlocking. When heat causes the bridge to expand during the sunlight hours of a hot day, the two sides of the expansion joint move toward one another; then, as the bridge cools down after dark, they begin gradually to retract. Thus the bridge has a built-in safety zone; otherwise, it would have no room for expansion or contraction in response to temperature changes. As for the use of the comb shape, this staggers the gap between the two sides of the expansion joint, thus minimizing the bump motorists experience as they drive over it.
Railroad tracks
Most railroad tracks are built from pieces of steel supported by wooden ties, and laid with a gap between the ends. This gap provides a buffer for thermal expansion, but there is another matter to consider: the tracks are bolted to the wooden ties, and if the steel expands too much, it could pull out these bolts. Hence, instead of being placed in a hole the same size as the bolt, the bolts are fitted in slots, so that there is room for the track to slide in place slowly when the temperature rises.
Power lines
Another example of thermal expansion in a solid is the sagging of electrical power lines on a hot day. This happens because heat causes them to expand, and, thus, there is a greater length of power line extending from pole to pole than under lower temperature conditions.
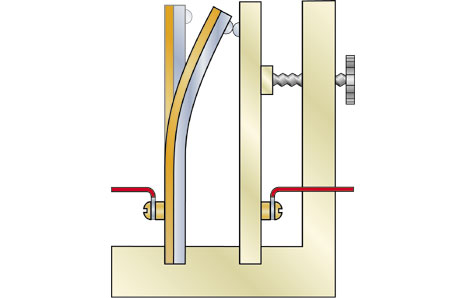
Bimetallic strips in thermostats
Diagram of a thermostat in an air-conditioning unit. A thermostat is used to regulate and maintain temperature. The bimetallic strip below bends according to the temperature, and becomes more bent as its heat increases. If it becomes so hot and bent that it touches the plate on the right, it completes an electric circuit, which turns the air-conditioning unit on. As the air-conditioning cools the surrounding air, the metallic strip also cools, causing it to straighten and break the circuit.
5.2 Thermal Capacity
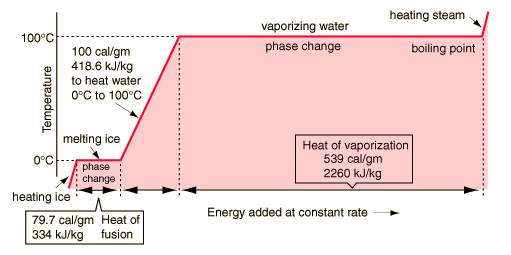
If you heat a piece of ice at, say, -10°C, its temperature rises until it reaches 0°C and then remains constant until all the ice has melted. The temperature then starts to rise again.The energy supplied during the period when the temperature was constant is called latent heat.
Latent heat is the 'hidden heat' used to change the state of a substance without a change in temperature.Latent heat means ‘hidden’ or concealed energy, in the sense that it does not show up on a thermometer.
Usually, when a substance is heated, its temperature rises. However, when matter changes phase from solid to liquid, or liquid to gas, there is no actual temperature rise. The energy is used to overcome the forces of attraction, which hold the water molecules together in the solid state (ice) so that they become relatively free to move.
Latent Heat of Vaporisation: The change of phase from liquid to gas
Latent heat of fusion: The change of phase from solid to liquid.
Latent Heat: Amount of energy associated with phase change (kJ/kg)
The formula for Specific heat capacity is:
Q = ml
Q- amount of energy released or absorbed during the change of phase (in kJ )
m- is the mass of the substance (in kg),
L- is the specific latent heat for a particular substance (kJ/kg)
The specific latent heat of fusion of ice is 336 KJ kg –1.The specific latent heat of fusion of ice is the amount of heat required to change 1 kg of ice to water without a change in temperature.
The specific latent heat of vaporisation of water is 2260 KJ kg –1.The specific latent heat of vaporisation of water is the amount of heat required to change 1 kg of water to steam without a change in temperature.
The formula for Specific heat capacity is :
E = m x c x ΔT
we can rearrange the equation to get
c=E/ m x ΔT
We want to find the Specific Heat capacity of water, which we know to be around 420KJKg-1°C-1
Latent heat is the 'hidden heat' used to change the state of a substance without a change in temperature.Latent heat means ‘hidden’ or concealed energy, in the sense that it does not show up on a thermometer.
Usually, when a substance is heated, its temperature rises. However, when matter changes phase from solid to liquid, or liquid to gas, there is no actual temperature rise. The energy is used to overcome the forces of attraction, which hold the water molecules together in the solid state (ice) so that they become relatively free to move.
Latent Heat of Vaporisation: The change of phase from liquid to gas
Latent heat of fusion: The change of phase from solid to liquid.
Latent Heat: Amount of energy associated with phase change (kJ/kg)
The formula for Specific heat capacity is:
Q = ml
Q- amount of energy released or absorbed during the change of phase (in kJ )
m- is the mass of the substance (in kg),
L- is the specific latent heat for a particular substance (kJ/kg)
The specific latent heat of fusion of ice is 336 KJ kg –1.The specific latent heat of fusion of ice is the amount of heat required to change 1 kg of ice to water without a change in temperature.
The specific latent heat of vaporisation of water is 2260 KJ kg –1.The specific latent heat of vaporisation of water is the amount of heat required to change 1 kg of water to steam without a change in temperature.
The formula for Specific heat capacity is :
E = m x c x ΔT
we can rearrange the equation to get
c=E/ m x ΔT
We want to find the Specific Heat capacity of water, which we know to be around 420KJKg-1°C-1
5.3 Melting and boiling
There are two types of energy in a substance, which we often call to be the internal energy. The two types of energy are potential and kinetic energy.
The kinetic energy is what defines the temperature of the substance. When the temperature of a substance reaches a state where it is ready to go through boiling/melting, energy is still being transferred but the temperature is still constant.
In this process, bonds are being broken/formed, so energy transfer is definitely required for this change in state. However, if the kinetic energy doesn’t change here, what changes? The potential energy increases.
Boiling and evaporation
One difference between boiling and evaporation is that evaporation happens at the surface of a liquid where boiling occurs inside the body of the liquid.
Additionally, evaporation can literally happen at any temperature, as long there is enough Kinetic Energy to break bonds, but boiling can only happen at certain temperatures, e.g. 100°C for water.
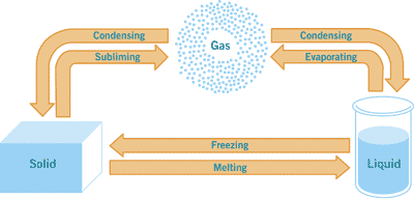
Condensation and Solidification
Condensation: This is the change of the physical state of matter from a gas into a liquid.
Basically, condensation starts by the formation of atomic clusters of the substance in its gaseous (gas) state, like the formation of a rain drop in the clouds.
Solidification: This is the change of the physical state of matter from a liquid to a solid.
A good example is changing water into ice cubes. You put the water into the fridge, and it freezes to become ice
The kinetic energy is what defines the temperature of the substance. When the temperature of a substance reaches a state where it is ready to go through boiling/melting, energy is still being transferred but the temperature is still constant.
In this process, bonds are being broken/formed, so energy transfer is definitely required for this change in state. However, if the kinetic energy doesn’t change here, what changes? The potential energy increases.
Boiling and evaporation
One difference between boiling and evaporation is that evaporation happens at the surface of a liquid where boiling occurs inside the body of the liquid.
Additionally, evaporation can literally happen at any temperature, as long there is enough Kinetic Energy to break bonds, but boiling can only happen at certain temperatures, e.g. 100°C for water.
Condensation and Solidification
Condensation: This is the change of the physical state of matter from a gas into a liquid.
Basically, condensation starts by the formation of atomic clusters of the substance in its gaseous (gas) state, like the formation of a rain drop in the clouds.
Solidification: This is the change of the physical state of matter from a liquid to a solid.
A good example is changing water into ice cubes. You put the water into the fridge, and it freezes to become ice