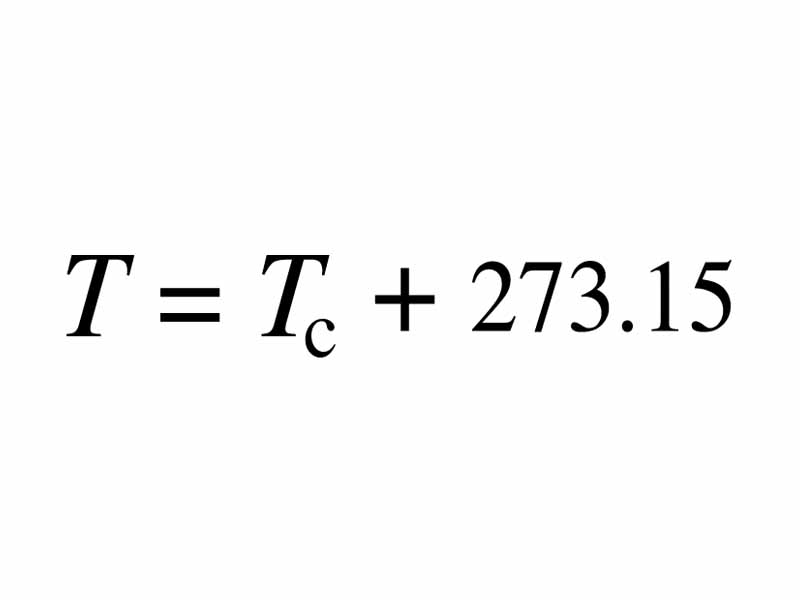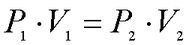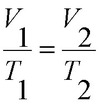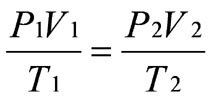P4. Simple kinetic molecular model of matter
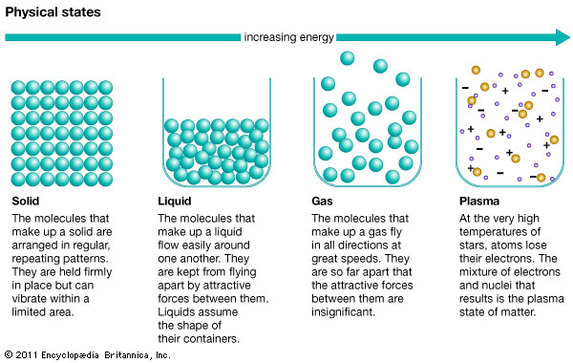
4.1 States of matter
4.2 Molecular Model
| Kinetic Theory Class Worksheet | |
| File Size: | 493 kb |
| File Type: | |
The molecular model uses this idea that all materials are made up of atoms that behave rather like tiny spheres.
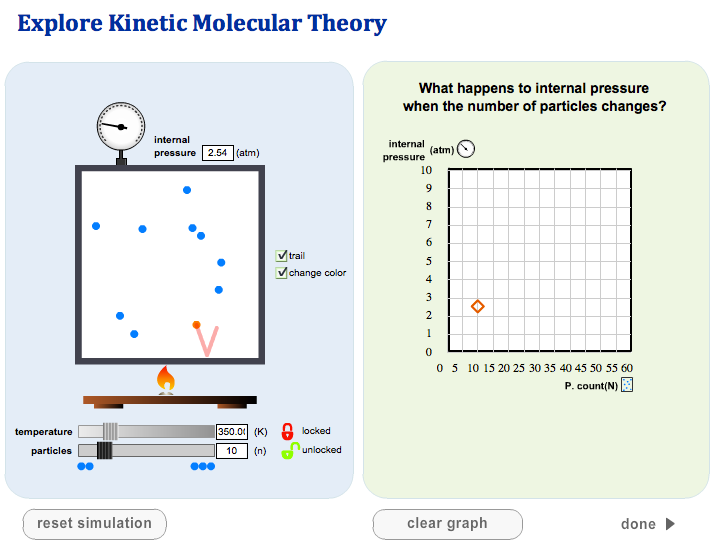
The molecular theory shows how individual particles interact with one another. The molecular theory contains five general postulates (A thing suggested or assumed as true as the basis for reasoning, discussion, or belief):
The molecular theory shows how individual particles interact with one another. The molecular theory contains five general postulates (A thing suggested or assumed as true as the basis for reasoning, discussion, or belief):
- Molecules are point masses (they have no volume)
- Molecules exert no force on each other unless they collide
- Collisions of molecules with each other or the walls of the container do not decrease the energy of the system
- The molecules of a gas are in constant and random motion
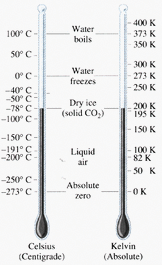
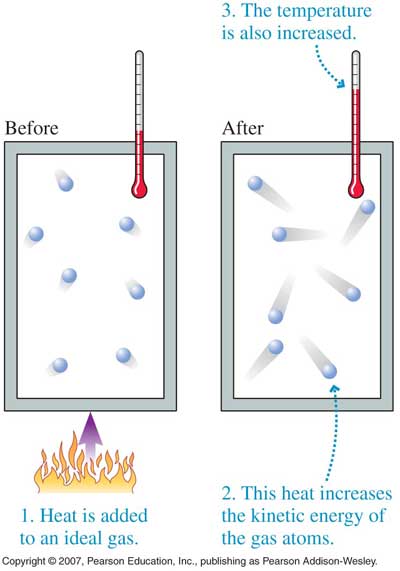
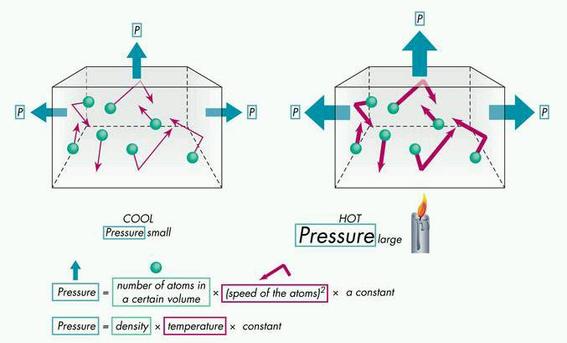
- The temperature of a gas depends on its average kinetic energyavg(1/2mv^2) = 3/2kT . In other words, the energy of an ideal gas is entirely kinetic.
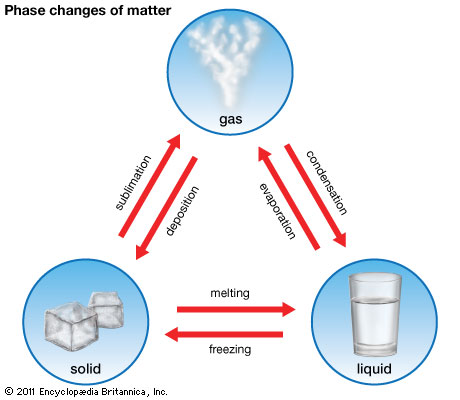
4.3 Evaporation
|
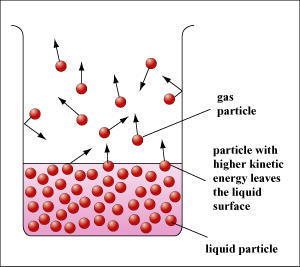
Inside a liquid, the distribution of kinetic energy of the particles is random and some has higher and some lower kinetic energies. The energetic particles have enough energy to overcome the attractive forces between them and leave the liquid through the liquid surface.
As the particles that leave the liquid are the high-energy particles, the average kinetic energy of the liquid particles decreases and thus the temperature of the liquid decreases. Thus the liquid cools down during evaporation. Since evaporation is a change of phase from liquid to gas, latent heat of vaporization is involved. In other words, the evaporated vapour takes away latent heat and cools down the liquid. |
|
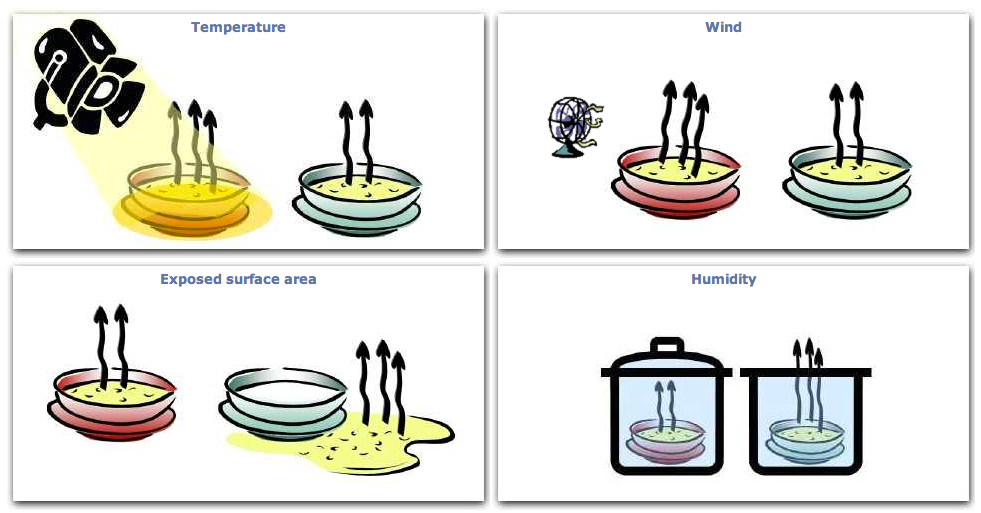
Factors affecting the rate of evaporation
The rate of evaporation increases if the temperature of the liquid is increased. It is also increased if:
The rate of evaporation increases if the temperature of the liquid is increased. It is also increased if:
- the surface area of the liquid is increased
- air is moving over the surface of the liquid.
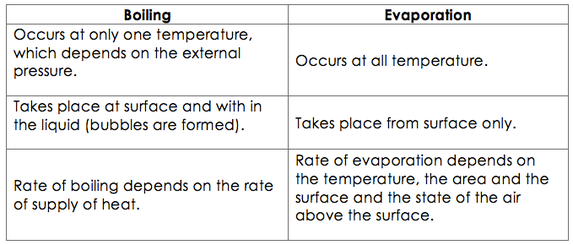
Difference between boiling and evaporation
4.4 Pressure changes
| Balloon in the flask demonstration | |
| File Size: | 236 kb |
| File Type: | |
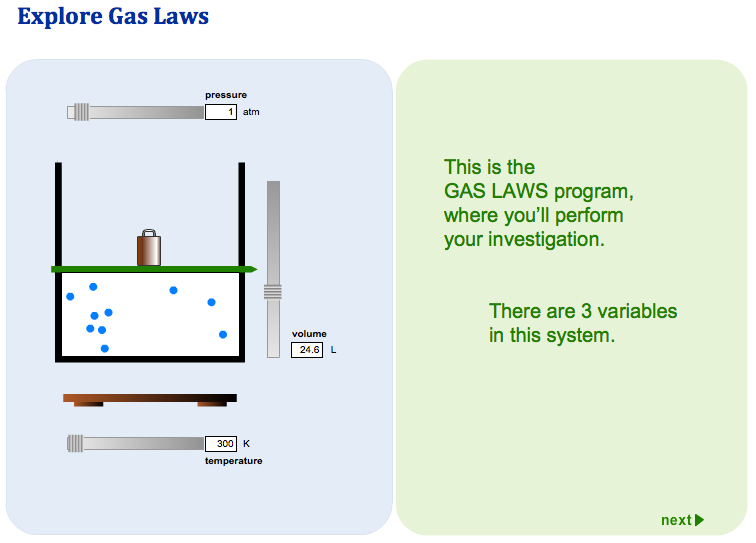
The Gas Laws
| Gas Law Class Worksheet | |
| File Size: | 186 kb |
| File Type: | |
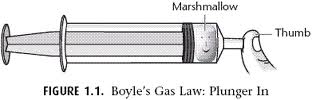
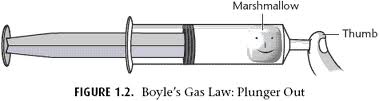
Boyle's Gas Law: Marshmallow Under Pressure Lab
| Marshmallow under pressure lab | |
| File Size: | 101 kb |
| File Type: | |
|
Step 1
|
Step 2
|
The Collapsing Fizzy Drink Can
| Collapsing fizzy drink can lab | |
| File Size: | 75 kb |
| File Type: | |
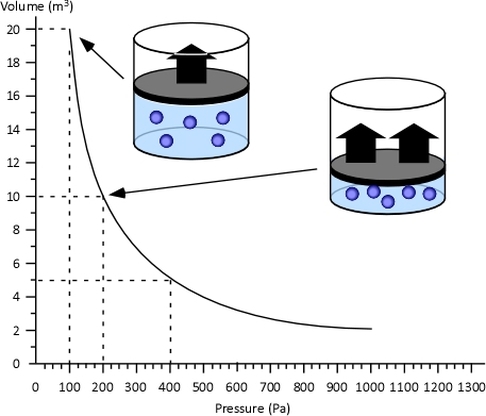
Boyle’s Law: Boyle discovered that as pressure on a gas was increased, its volume decreased. Boyle’s Law states that if the temperature of an ideal gas is held constant, the pressure and volume of a given amount (mass or number of molecules) of an ideal gas are inversely proportional, as pressure increases, the volume occupied by the gas decreases.
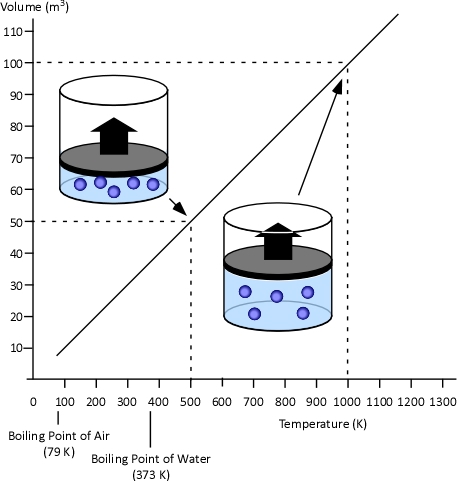
Charles’s Law: Charles found that as the gas was heated, the volume increased. As the gas was cooled, the volume decreased. This finding gave rise to Charles’s Law which states that at a constant pressure the volume of a given amount (mass or number of molecules) of an ideal gas increases or decreases in direct proportion with its absolute (thermodynamic) temperature.
Revision
Simple Kinetic Molecular Model revision site